The pharmaceutical company Merck recently announced that, according to unpublished research, a drug called molnupiravir may reduce the risk of hospitalization or death from COVID-19 by 50%. If true, this would be a remarkable result. The US federal government has apparently pre-purchased a large number of doses in case the drug ends up being approved for widespread use by the FDA.
Molnupiravir is just the latest antiviral drug being touted as a potential way to treat or help prevent COVID. Other antivirals, such as remdesivir, hydroxychloroquine, and ivermectin have also been touted as potential COVID drugs despite limited or mixed evidence for their effectiveness. They have generally become highly politicized, with partisan squabbles drowning out measured assessments of what the evidence does and does not say about their known utility for COVID.
Below I will describe the difference between antiviral drugs and vaccines, what we seem to know about this new Merck drug, and point out some cautionary observations that aren’t getting much attention in mainstream media sources.
Antiviral Drugs vs. Vaccines
Vaccines train your immune system to recognize a pathogen. Viral vaccines contain either a killed or weakened version of a virus, or, in the case of the new Moderna and Pfizer vaccines, an mRNA encoding a specific virus protein. When put into the body, these do not trigger a full-blown infection but are nonetheless recognized by our immune systems, which can learn and remember what they encounter. This allows us to mount an immune response in the future if we are infected by the real pathogen. This “pre-training” of the immune system can last for months or years after vaccination depending on the vaccine and the nature of the pathogen they’re designed for.
Antiviral drugs, by contrast, are only effective while the drug is in your system. They directly interfere with some aspect of virus biology, such as the ability to replicate within our cells. They are sometimes taken on an ongoing basis to help prevent an infection from taking root, or, in the case of COVID drug Merck is advertising, given during an infection to help reduce its severity. But once the drug is out of your system, there’s no lasting effect.
Antiviral drugs and vaccines are not necessarily alternatives to one another. They can both be used in a complementary fashion in order to minimize the odds of contracting an infection or experiencing severe illness.
Merck’s Drug Molnupiravir: how does it work?
Molnupiravir is a prodrug, which means our bodies metabolize it into an active drug that actually does something. This is common. In this case, the active drug has the unfortunate name of β-D-N4-hydroxycytidine triphosphate. Let’s just call it Merck’s COVID drug. It’s a “ribonucleoside analog,” which means its chemical structure resembles one of the base pairs in RNA. Recall that SARS-CoV-2 is an RNA virus: it’s genome is composed of RNA, not DNA.
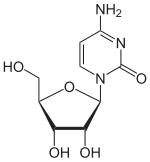
The resemblance of Merck’s drug to an RNA base pair is related to its mechanism of action. When an RNA virus like SARS-CoV-2 tries to replicate in the drug’s presence, it uses the drug rather than the appropriate molecule in the replication process. This can result in widespread mutations, which can destroy to virus’ ability to replicate altogether.
If a drug like this is good at causing catastrophic mutations for the virus (and does not cause serious collateral damage), it could be used prophylactically to help prevent an infection, or else as a treatment during an ongoing infection to reduce its severity.
Merck’s Drug Molnupiravir: cautionary notes
Given the scale of the ongoing COVID-19 pandemic, people are obviously eager for any new treatment or preventative measure we can come up with. One the one hand, we want to act quickly to minimize the spread of the virus. On the other hand, we shouldn’t allow ourselves to become careless and overlook potential downsides to a new treatment.
Given that the US government has already pre-purchased millions of doses of Merck’s drug and the FDA will presumably look to fast-track its approval, here are two things we should not lose site of:
1.) There is in vitro research indicating that Merck’s drug may also cause mutations in mammalian cells. When applied to dividing cells growing in a petri dish, Merck’s drug not only causes debilitating mutations for the virus but can also induce mutations in the mammalian host cells the virus is replicating within. If this drug were to cause such mutations when given to a human, it could contribute to the development of cancer or birth defects.
We need to carefully determine what the mutation risk to our own cells might be at different doses of this drug compared to the strength of its antiviral effects at each dose. Ideally, there will be a dose range that is antiviral but poses no significant risk of mutagenesis for our own cells. But there are no guarantees. We should work this out before fast-tracking the drug for widespread human consumption.
2.) Non-lethal mutations could conceivably facilitate the evolution of more new SARS-CoV-2 variants. I was curious about this when I learned that Merck’s drug worked by causing mutations. If the drug causes mutations for the virus, is there any risk of it causing beneficial mutations for the virus, leading to the evolution of yet more SARS-CoV-2 variants?
I asked population geneticist Dr. Sarah Otto about this, who I had previously spoke to about the evolution of SARS-CoV-2 variants.
Again, we should proceed with great care. Merck’s drug may indeed have potent antiviral activity against SARS-CoV-2 and other RNA viruses, but that beneficial property needs to be balanced against the risk of the drug having undesirable consequences, such as facilitating the evolution of more new variants.
The crux of the problem: research takes time. There is a tradeoff between acting quickly to help fight the pandemic ASAP, and investing more time in additional research to better understand all of the potential downstream consequences widespread use of such a drug might have.
Not an easy problem.
Controversy Surrounding Other Antiviral Drugs
There have been at least three other antiviral drugs in the headlines in the past year, which I will only touch on briefly.
Remdesivir: a broad-spectrum antiviral developed by Gilead Sciences. The FDA approved its use in the US in October 2020, with mixed evidence of its effectiveness at helping speed recovery from COVID—some studies showing it helped, and others that it had no statistically significant effect.
Hydroxychloroquine: This drug was already used to help treat or prevent things like malaria and rheumatoid arthritis. There is mixed evidence that it may be helpful with COVID-19, with results likely highly dependent on the dose used and how early after initial infection it is given. Highly politicized.
Ivermectin: This drug, originally marketed by Merck in the 1980s, has been widely used around the world for decades to treat parasite infestations in both humans and animals. Its effectiveness as an anti-parasitic led to its discoverers winning the 2018 Nobel Prize.
In vitro studies have indicated it has antiviral activity against several different RNA viruses. There has been mixed evidence for its effectiveness against COVID-19, with some indications or benefit, some of no significant effect, and debate surrounding the quality of existing research. The use of this drug as a prevention measure for COVID has become ultra-politicized.
Unlike some other drugs, ivermectin is cheap and is not known to be toxic or mutagenic in humans when used at prescribed doses. We will know more about its real COVID effectiveness once higher-quality, randomized trials are finished.
Merck’s Drug Molnupiravir: best and worst case scenarios
The best case scenario for the new Merck drug is that clinical trials confirm it has robust antiviral activity against COVID-19 for humans, we additionally confirm that the doses needed for this effect do not cause mutations in our own DNA, and that there’s little risk of drug-induced mutations facilitating the evolution of new SARS-CoV-2 variants.
The worst case scenario is that this drug is rushed into widespread human use because of its antiviral properties without the proper research to confirm the other risks. If it also induces mutations in our own cells, the consequences of this would likely not be known or felt for years, long after it was too late. If it also facilitated the evolution of new SARS variants, that may also be difficult to discern amidst the full complexity of the ongoing pandemic, with multiple new variants already spreading.
I will be watching closely to see if any new research is happening to confirm whether this drug has the ability to induce mutations in human cells and how quickly it speeds through regulatory approval processes.
No matter what, it’s important not to overlook the obvious: diet and exercise are critical for preserving health and preventing disease. Antiviral drugs can and should be use when there is clear evidence of benefit and minimal risk of harm. But we can’t pin our hopes on magic pills being developed so that we can passively avoid getting sick. We must actively preserve our health every day. Pill or no pill, most of us can be healthier by simply being more active.